The spectrum of the food on offer is extending because of the globalisation
of world trade. This also applies to tropical fruits, so the consumer is
increasingly into contact with them. The annual world production of 14.7
Mio tons makes mango (Mangifera indica) one of the most popular
tropical fruits. World-wide it is cultivated in approximately 1000 strains.
In Europe just a few strains are sold, because most of them taste petroleum-like
and are not accepted for that reason. Tommy Atkins, which is cultivated
particularly in South America, is the variety most frequently sold. Other
well known strains are Osteen (Spain), Eden (Israel) and Ngowe (Kenya)
(Franke 1997).
Allergic reactions to mango have become increasingly important. Sensitizations
to mango pollen (Vargas Correa et al. 1991), seed (Fernandez et al. 1995)
and fruit were described, respectively. In a French study it could be proved
that approx. 6 % of the examined persons with clinically manifest food
allergy showed a sensitization to mango (Andre et al. 1994). Moreover ingestion
of mango fruit caused anaphylactic reactions in several cases (Dang &
Bell 1967, Wüthrich & Hofer 1984, Miell et al. 1988, Jansen et
al. 1992). Mango fruit allergens are reported to cross-react with latex
allergens (Brehler et al. 1997), mugworth allergens (Wüthrich &
Hofer 1984), peanut allergens (Fernandez et al. 1995), and birch pollen
allergens (Wellhausen et al. 1996).
In some fruits a variety dependence of their allergenic potency is described.
For example, apple-allergic patients frequently report that some apple
strains usually are highly allergenic (i.e. Granny Smith, Golden Delicious)
whereas others (i.e. Jamba, Gloster, Boskop) are tolerated without or with
moderate symptoms (Vieths et al. 1993, 1994). With regard to the described
phenomena, the aim of the present investigation was to study differences
in the IgE-binding patterns and in the allergenicity of fruit allergens
in four different mango varieties.
Patients' sera
Seven sera were obtained from patients with RAST classes
> 3 for specific IgE to mango by CAP-RAST (Pharmacia, Uppsala, Sweden).
All patients were adults (three female and four male), aged between 19
and 59 years. The sera were collected at the University Hospital Eppendorf,
Department of Dermatology and Allergy, Hamburg, Germany. The pooled serum
contained equal aliquots of the sera.
Mango Extracts
The mango varieties were purchased from Fa. Inter, Großmarkt
Hamburg, Germany. All fruits were in a consumable condition and harvested
five days before preparing the allergen extracts. They were prepared by
a low-temperature extraction method, as described previously but with some
minor modifications (Möller et al., 1997a, 1997 b). They were freeze-dried
and stored at 20 °C until use. The protein concentration was determined
using a Bradford (1976) assay.
Chemicals
If not otherwise mentioned, all chemicals were of analytical
grade.
SDS-PAGE
Discontinuous SDS-PAGE was performed according to the
method of Laemmli (1970) with a 5 % (w/v) acrylamide stacking gel and a
13 % (w/v) acrylamide resolving gel. Protein samples were reduced with
beta-mercaptoethanol (sample buffer according to Vieths et al. 1992) and
loaded onto the gel at a concentration of approx. 10 µg/cm. The gels
(250 x 120 x 0.5 mm) were developed in a horizontal slab gel apparatus
(Desaphor HF, Desaga, Heidelberg, Germany). Gels were silver stained according
to Morissey (1981).
Immunoblotting
For immunoblotting approximately 10 µg protein/cm
were loaded onto the gels. Protein transfer was carried out as described
previously (Möller et al. 1997a, 1997b). Immunostaining was performed
according to Vieths et al. (1992) with minor modifications. The dried nitrocellulose
membranes were blocked twice (15 min) with blocking buffer consisting of
5 % skimmed milk powder, and 0.1 % Tween 20 in phosphate buffered saline
[PBS (0.01 M potassium phosphate buffer, pH 7.4, 0.13 M sodium chloride)].
Nitrocellulose strips were incubated overnight with 100 µL of patients
sera in 1400 µL incubation buffer [0.3 % bovine serum albumin (BSA),
0.1 % Tween 20 in PBS]. After incubation with rabbit anti-human IgE (1:4000,
60 min), biotinylated goat anti-rabbit IgG (1:6000, 60 min; both reagents
from Dako, Copenhagen, Denmark) and streptavidin-horseradish peroxidase
conjugate (1:20000, 20 min, Medac, Hamburg, Germany), the blots were stained
with 3,3',5,5'-tetramethylbenzidine-dioctylsodiumsulphosuccinate (TMB).
One strip was incubated with serum from a non allergic
patient to check non-specific binding. To check the efficiency of protein
transfer, one strip with separated extract and one strip with low molecular
weight marker proteins (Pharmacia) were stained with colloidal gold (Bio
Rad).
Immunoblot Inhibition
A total of 100 µL of the pooled serum was incubated
with 100 µg of protein of the protein extracts in a total volume
of 1.5 mL of incubation buffer for 60 min at room temperature. Afterwards,
the preincubated solution was immunostained as previously described
(Möller et al. 1997a, 1997 b).
Enzymeallergosorbent test (EAST) Inhibition
Protein extracts were diluted to total protein concentration
of 10 mg/L using a sodiumcarbonate-buffer (50 mM NaCO3,
pH 9.6). 300 µL were added into each cavity of a Maxisorb-Mikrotiterplate
(Nunc, Roskilde, Denmark) and coupled over night at room temperature. Free
binding sites were blocked with 5 % skimmed milk powder and 0.1 % Tween
20 in PBS for 1 h. Pooled serum was diluted 1:2 in incubation buffer (0.3
% BSA, 0.1 % Tween 20 in PBS). A dilution series of the inhibitor-extracts
was prepared in six steps using the same incubation buffer (undiluted,
1:10, 1:100, 1:500, 1:1000, 1:10000). Ovalbumin was used to check non-specific
inhibition. A total of 120 µL of diluted poolserum was preincubated
with 120 µL of the inhibitor solution for 1 h. 50 µL of this
solution were put into the cavities of the microtiter plate. It was incubated
overnight at room temperature in the dark. After three washes with 1 %
Tween 20 in PBS, 50 µL of anti-human IgE alkaline phosphatase conjugate
(Allergopharma, Reinbek, Germany), diluted 1:200 in incubation buffer,
were added and incubated overnight. The plates were washed again and the
bound enzyme activity was stained with p-nitrophenylphosphate (PNPP) for
60 min at 37 °C using an alkaline phosphatase staining kit (Allergopharma).
Absorbance was measured at 405 nm.
Silver Staining
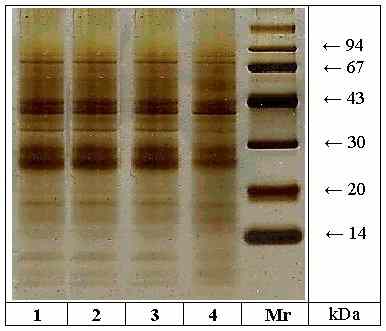
Extracts of the four mango varieties were investigated by SDS-PAGE
/ silver staining (Fig. 1). In all varieties proteins with Mr between 14
and 94 kDa were detected. All strains showed three additional protein bands
< 14 kDa. Between 14 and 20 kDa three protein bands with Mr of approximately
15, 17 and 19 kDa were detected, at the same time the protein bands with
Mr of 15 and 17 kDa were most intense in the strain Ngowe. Very strongly
detected proteins became apparent at Mr of approx. 27 and 29 kDa in all
four strains. The protein band with Mr of 35 kDa was most intense in the
strains Osteen, Eden and Tommy Atkins. At 40 and 43 kDa there were additional
strongly detected proteins. Between 43 and 67 kDa there was just a diffuse
background of unresolved proteins. At approximately 70 kDa there were two
distinct protein bands. In conclusion there were no significant differences
in the protein pattern between the four investigated strains.

Figure 1: SDS-PAGE / silver staining
of protein extracts from different mango varieties. Lane
1: Osteen, lane 2: Eden, lane 3: Tommy Atkins, lane 4: Ngowe (Mr = molecular
mass marker proteins).
Immunoblotting
The allergens in extracts of the mango varieties Osteen, Eden, Tommy
Atkins and Ngowe were characterized by SDS-PAGE / immunoblotting using
seven sera from mango allergic patients.
All sera detected the same allergens in each strain. The detection
of the four varieties with pooled serum is shown in Fig. 2. Allergens with
Mr of approx. 14, 30, 40, 43 and 67 kDa were visible in each strain (Lane
1-4). Furthermore there was no difference between the varieties in detection
intensity of any allergen.
Immunoblot-Inhibition
Cross-reactivity among the different mango varieties was determined
by means of immunoblot-inhibition. Fig. 3 shows the separated extract from
strain Eden. Preincubation of the pooled serum with mango extracts (incl.
Eden itself) resulted in total inhibition of IgE binding by all four strains.
No significant inhibition or non-specific binding was observed with ovalbumin
and with the control serum, respectively.
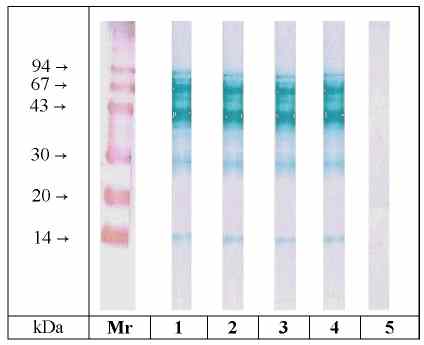
Figure 2: SDS-PAGE / Immunoblotting of different mango varieties. Lane 1: Osteen, lane 2: Eden, lane 3: Tommy Atkins, lane 4: Ngowe, lane 5: Osteen / detection with control serum. Lanes 1-4 were detected with pooled serum from mango- sensitive patients. (Mr = molecular mass marker proteins, colloidal gold staining)
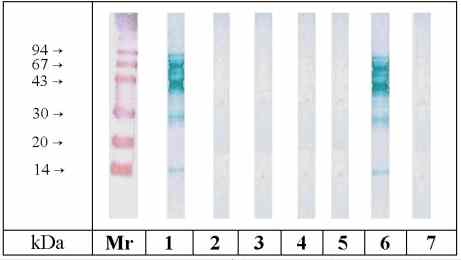
Figure 3: Immunoblot-Inhibition of IgE binding to SDS-PAGE
separated mango Eden extract. Inhibitors (100 µg
protein / inhibition): Lane 1: no inhibitor, lane 2: Osteen, lane 3: Eden,
lane 4: Tommy Atkins, lane 5: Ngowe, lane 6: non specific inhibition with
ovalbumin, lane 7: control serum / no inhibitor. All inhibitors were preincubated
with pooled serum from mango-sensitive patients. (Mr = molecular mass marker
proteins, colloidal gold staining)
EAST Inhibition
In order to estimate the extent of cross-reactivities among the mango
varieties EAST inhibition experiments were performed using the pooled serum.
The inhibition of IgE binding to the adsorped protein extract from Mango
Osteen is shown in Fig. 4 (Figures of other strains are not shown since
they show almost the same results). All four inhibition graphs were nearly
identical. The protein concentrations resulting in a 50%-inhibition of
IgE binding are given in Table 1. For example IgE binding to Mango Osteen
was strongly inhibited by preincubation with all other strains and vice
versa. 50%-inhibition values were in the range of 0.5 to 1.4 µg/ml.
Using the post-hoc comparison technique there was no difference between
the mean values of 50 % inhibition of different strains (p<0.05). Total
inhibition of IgE binding was achieved by all four varieties at concentrations
below 100 µg/mL. No significant inhibition was observed with ovalbumin
in all cases.
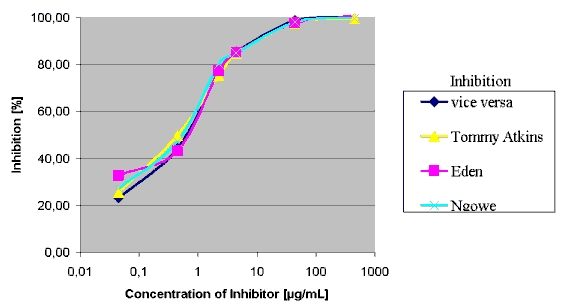
Figure 4: EAST-Inhibition of IgE-binding to mango strain Osteen.
Inhibitors: Osteen, Eden, Tommy Atkins, Ngowe.
Table 1: EAST inhibition with different mango extracts. The
protein concentration [µg/mL] for 50 % inhibition is given.
| Inhibitor | ||||
| Coupled allergen | Osteen | Eden | Tommy Atkins | Ngowe |
| Osteen | 0.7 | 0.8 | 0.5 | 0.6 |
| Eden | 0.9 | 0.5 | 0.6 | 0.8 |
| Tommy Atkins | 0.5 | 0.5 | 1.0 | 1.4 |
| Ngowe | 1,3 | 0,7 | 0,8 | 0,8 |
Investigations of different apple strains showed major differences in the allergenic potency of these strains (Vieths et al. 1993, 1994). Because the history of breeding and cultivation of mangos is just as extensive as in the case of apples, the present study was undertaken to prove whether there are variety dependent differences of the allergenic potency of mango fruit or not. For this purpose the four most consumed mango strains, which were cultivated in four different continents (Africa, Asia, Europe and South America) were selected.
The silver staining showed no significant difference between the varieties. These findings were confirmed by immunoblotting. In all strains allergens with Mr of approximately 14, 30, 40, 43 and 67 kDa were detected. Due to the fact that the intensity of the allergen bands seemed to be nearly identical it can be assumed that there are no obvious differences in the IgE binding patterns of mango fruit allergens in the four strains. This result could be confirmed by means of immunoblot inhibition. It was shown that homologous inhibition as well as inhibition with the other strains led to a complete inhibition, indicating the occurrence of identical allergens in all strains.
The results were confirmed by EAST inhibition. It could be shown that
all inhibition curves ran almost congruently. Significant differences were
not observed. Using statistical examination
by means of LSD test it could be stated that the average values of 50 %
inhibition do not differ significantly from each other.
Thus it was demonstrated that the allergenic potencies of the four
most consumed mango strains in Europe do not deviate from each other.